Tag: FDA regulations
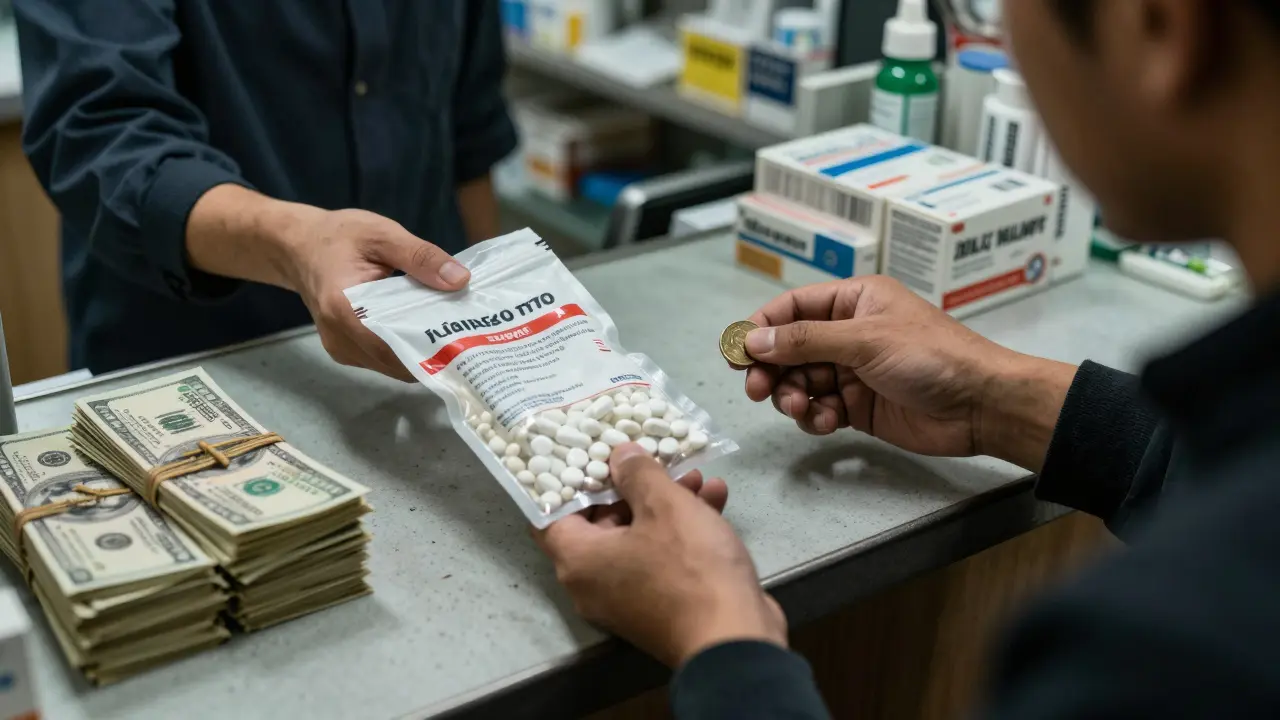
Generic Drug Price Trends: Year-by-Year Changes Explained
6 Feb
|
01:19 AM
Explore how generic drug prices change yearly due to competition, regulations, and market dynamics. Learn why some generics spike in cost while others drop, and what this means for patients and healthcare.
read morerecent posts

Quality Control Testing: A Step-by-Step Guide to Manufacturing QA
Learn the essential steps of quality control testing in manufacturing, from raw material inspection and IPQC to ISO standards and CAPA procedures.
Published ON: 4 Apr
Generic Competition Explained: How Second and Third Entrants Slash Drug Prices
Discover how additional generic drug manufacturers drive down costs. Learn why the second and third generic entrants are crucial for significant price reductions and why some drugs stay expensive.
Published ON: 1 Apr